| Contents | Previous | Next |
by Michael A. Siegel, DDS, MS
Abstract: Oral mucosal disorders are frequently encountered by the practicing dentist. These lesions may represent oral manifestations of dermatologic or systemic disease, reactive lesions, or occult neoplasms. The diagnosis of these conditions is usually based on case-specific historical findings, clinical appearance, and the results of diagnostic procedures. This article will discuss the diagnosis and management of commonly occurring oral mucosal conditions such as candidosis, recurrent aphthous ulceration, herpes virus infection, and lichen planus. This manuscript represents a synthesis of the literature and the management approach utilized by the author in the treatment of his patients. This article is not intended as a comprehensive review of all the subjects discussed.
Dentists are frequently called upon to evaluate oral mucosal conditions. This article will present the diagnosis of and management utilized by the author for four of the most frequently encountered oral lesions: candidosis, recurrent aphthous ulceration, herpetic infection, and lichen planus.
Candidosis, the most commonly occurring oral fungal infection in the non-immuno compromised patient, is caused by Candida albicans. Up to 60 percent of healthy individuals may harbor this fungal organism as host flora in the oral cavity1. For individuals who have Candida albicans as a normal component of their oral microflora, their own immune system and the competing bacteria keep the fungal organisms from overgrowing.2 This opportunistic infection may occur due to a variety of systemic factors or as a result of local changes in the oral environment.
Systemic conditions associated with the development of candidosis include systemic steroid therapy and endocrine disturbances such as diabetes, pregnancy, and hypoparathyroidism. Other systemic factors that may favor the development of candidosis include malabsorption and malnutrition, Sjogren’s syndrome, cancer chemotherapy, and immuno suppression such as seen in AIDS patients.3 Local factors that predispose the patient to developing candidosis include changes in the oral flora resulting from decreased tissue resistance due to xerostomia or chronic local irritants (dentures, orthodontic appliances, smoking) and antibiotic therapy.
 |
 |
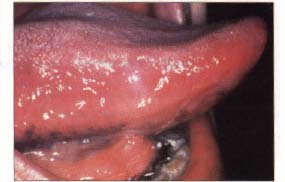
| Figure 1a. Pseudomembranous candidosis on the right side of the tongue in a 72-year-old male. | Figure 1b. Appearance of the tongue following two weeks of therapy with clotrimazole troches. |
 |
 |
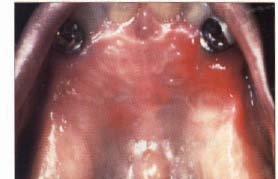
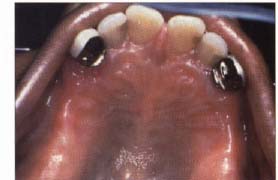
| Figure 2a. Erythematous candidosis of the palate in a 54-year-old female under an ill-fitting partial denture. | Figure 2b. Appearance of the palate following temporary reline of the denture and two weeks of therapy with nystatin ointment. |
The clinical appearance of the oral lesions can vary greatly. Pindborg reported four clinical varieties of oral candidosis found in HIV-infected individuals: pseudomembranous, erythematous, hyperplastic, and angular cheilosis.4 These clinical fungal subtypes are also useful in diagnosing and managing these lesions in nonHIV-infected individuals. The two most common oral presentations are pseudo membranous candidosis (thrush) and erythematous candidosis (denture sore mouth).
Pseudomembranous candidosis is characterized by the presence of white curd-like lesions that can easily be removed with a tongue blade or gauze, leaving an erythematous, erosive surface underneath (Figure 1). The term thrush should be reserved for use in a pediatric patient population. The white curds consist of fungal organisms, bacteria, inflammatory cells, fibrin, and desquamated epithelial cells. The lesions occur most commonly on the buccal mucosa and mucobuccal folds, the oropharynx, and the dorsal surface of the tongue. If the white lesions have not been rubbed off, the patients are usually asymptomatic. Patients with extensive erosive areas may complain of tenderness, burning, or dysphagia.
Erythematous candidosis is frequently noted in patients who wear maxillary complete or partial dentures, especially among those who do not remove the prosthesis prior to bedtime (Figure 2). This form of candidosis seems to occur as a result of decreased tissue resistance from the prosthetic appliance. The lesions have a distinct predilection for the palatal mucosa but may also occur under mandibular dentures. Clinically, erythematous candidosis primarily appears as red, atrophic lesions. The erythema may be diffuse, involving the entire denture-bearing area; or it may present as patchy areas of erythema that resemble petechiae.
Hyperplastic candidosis is unlike the pseudomembranous and erythematous forms in that it cannot be wiped off the mucosa. It is seen as elevated white plaques that resemble clinical leukoplakia. Hyperplastic candidosis most often involves the hard palate and the dorsal surface of the tongue. It must be distinguished from other keratoses by cytologic smear, culture, biopsy, or therapeutic trial with antifungal medication. Lesions that do not respond to a trial of antifungal medication must be biopsies to establish a diagnosis.
 |
 |
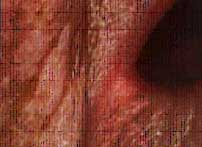 |
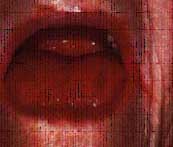
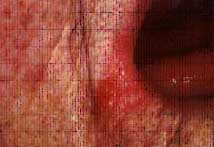
| Figure 3a. Severe bilateral angular cheilosis in a 76-year-old female patient. | Figure 3b. Close-up of the right commissure prior to therapy. | Figure 3c. Resolution of the right labial commissure following three weeks of therapy using nystatin with triamcinolone acetonide ointment. |
 |
 |
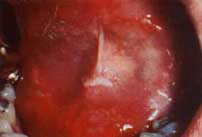 |
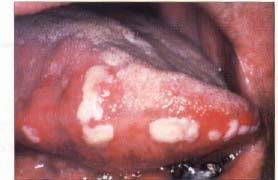
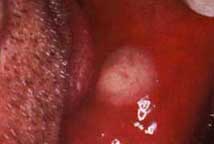
| Figure 4a. Minor recurrent aphthous ulceration of the upper lip. | Figure 4b. Major recurrent aphthous ulceration of the lower lip. | Figure 4b. Herpetiform recurrent aphthous ulcerations of the floor of mouth and ventral surface of the tongue. |
Angular cheilosis is due to candidal infection of the labial commissures. Angular cheilosis may occur with or without concurrent oral candidal lesions. It is characterized by redness and/or fissures radiating from one or both corners of the mouth and is often associated with small white plaques (Figure 3). Angular cheilosis has long been associated with Vitamin B deficiency and decreased occulding vertical dimension. While these conditions may serve as predisposing factors for the development of angular cheilosis, the management of the lesions must be directed at their fungal etiology.
| Table 1: Topical Medications Used to Treat Candidosis35 | ||||
|---|---|---|---|---|
| Medication | Forms | Dispense | Instructions | Advantages/Disadvantages |
| Nystatin | ointment | 30 gram tube. | Apply thin coat to inner aspect of denture(s) after each meal. Do not eat or drink for 30 min. following application. | Safe, high compliance, inexpensive. |
| Clolrimazole | troches | 70 | Dissolve slowly five times day until gone. Do not chew. | Safe, high pleasant tasting, high sugar content, expensive |
| Amphotericin | oral suspension 100 mg/ml | 48 ml | Swish with 1 ml for 3 min. four times daily and swallow until gone. | Topical and systemic effect, expensive, must remove dentures, potential side effects. |
| Nystatintriamcinolone acetonide | ointment | 15 gram tube | Apply to corners of mouth after meals, and at bedtime for two weeks. Do Not Lick. | Safe, inexpensive high compliance |
The diagnosis of candidosis can often be made from the patient history and the clinical appearance and distribution of the mucosal lesions. When necessary, especially in an immunocompromised individual, identification of the organisms is made in a culture of the lesion on a selective medium available from a commercial or hospital medical laboratory service, from a cytology smear stained with periodic acid Schiff (PAS) reagent, or on a wet smear macerated with 10 percent potassium hydroxide.
Oral candidosis is most often treated with topical antifungal agents such as nystatin ointment, clotrimazole troches, or amphotericin oral suspension (Table 1). Oral preparations in the form of troches provide the advantage of prolonged contact of the medication with the lesions. They are safe to use because of their poor systemic absorption. Oral hygiene must be reinforced when prescribing oral antifungal troches because of their sugar content. The sugar content of these medications can also present a problem when prescribed for diabetic patients on a strict carbohydrate diet.
While nystatin suspension is frequently prescribed, in general, rinses are less effective than other forms of topical antifungal therapy because the duration of tissue contact is insufficient.5 When a rinse must be prescribed, such as for xerostomic patients who cannot dissolve the clotrimazole troches, amphotericin oral suspension is preferred to nystatin suspension. When swallowed, amphotericin suspension provides both a topical acid systemic effect. However, its reported side effects include headache, nausea, vomiting, fever, and chills. Patients who wear dentures must remove them prior to using an antifungal rinse or troche.
When a dentist treats cases of erythematous candidosis that appear under a denture, the prosthetic appliance must be addressed as well as the oral lesions. After each meal, nystatin ointment or clotrimazole cream should be applied to the tissue side of the denture before replacement in the mouth. Patients should be reminded to remove their dentures at bedtime and soak them overnight in an antifungal solution. Most commercially available denture soaking tablets are fungicidal; it should not be necessary to prescribe nystatin suspension specifically for this purpose.
Angular cheilosis is a mixed infection of Candida albicans and salivary species of streptococci. These lesions respond very well to combination therapy containing an antifungal and a topical steroid in a cream or ointment vehicle. Nystatin with triamcinolone acetonide or clotrimazole with betamethasone dipropionate preparations are useful for this purpose. Patients should be discouraged from licking the lesions as this will continue to superinfect the cheilosis with salivary bacteria.
In cases of refractory candidosis, mucocutaneous candidosis, patients in whom compliance is a problem, or women who have a concurrent candida vaginitis, a systemic antifungal therapy with ketaconazole or fluconazole is recommended (Table 2). If either of these medications is used for longer than two weeks, liver function tests should be performed to watch for hepatotoxicity.
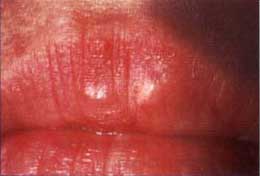 |
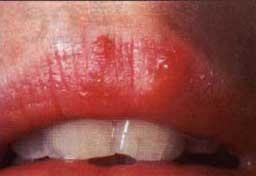 |
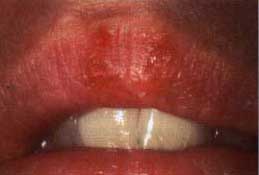
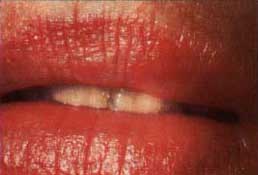
| Figure 5a. Recurrent herpes labialis in a healthy 40-year-old female. Early vesicle. | Figure 5b. Late vesicle. |
 |
 |
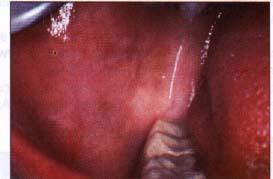
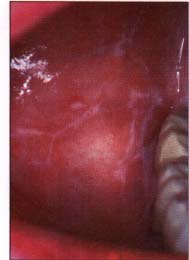
| Figure 5c. Crusted lesions. | Figure 5d. Healing without presence of scarring. |
 |
 |
| Figure 6a. Asymptomatic reticular lichen planus in a 43 year old male. Striae of Wickham of the right buccal mucosa. | Figure 6b. Resolution of the striae following two weeks of topical therapy with 0.05 percent fluocinonide cream. |
Recurrent aphthous ulcers, or canker sores, are the most commonly occurring nontraumatic ulcerations of the oral cavity, with the incidence rate varying from 20 percent to 60 percent of the population.6-7 Although the etiology of recurrent aphthous ulcers is still unknown, current investigations favor an immunologic reaction in which there is a focal immune dysfunction involving the lymphocytes8.
A number of factors play a modifying or triggering role in the development of these ulcers. These include hormonal changes, trauma, stress, and food allergies.9-10 Foods associated with triggering recurrent aphthous ulcers include bovine milk protein, glutens, chocolate, nuts, cinnamon, spices, and preservatives.11 A number of medications are known to cause these ulcers. The most commonly used medications reported to cause intraoral aphthous like lesions are the nonsteroidal antiinflammatory drugs.12 Deficiencies of ferritin and vitamin B12 have also been associated with recurrent aphthous ulcers. 13
These ulcers have long been associated with inflammatory bowel diseases. Inflammatory bowel disease may have extra-abdominal intraoral signs such as aphthous-like ulcers that may appear one year prior to radiographic abdominal changes.14 However, there is controversy as to whether recurrent aphthous ulcers represent a primary manifestation of granulomatous bowel diseases or a result of the medical management of these conditions.15
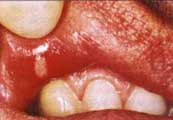
Recurrent aphthous ulcers have a characteristic clinical appearance of a shallow round or ovoid ulceration with a whitish center and an erythematous border. Clinically, recurrent aphthous ulcers present as single or multiple shallow ulcerations that occur on freely movable mucous membranes such as the lips, buccal mucosa, tongue, soft palate, floor of the mouth, and tonsillar pillars. They are categorized as minor, major, or herpetiform, depending on their size and duration (Figure 4).
Minor ulcers heal within two weeks and are less than or equal to 0.5 centimeters in diameter. Major ulcers are larger than 0.5 centimeters in diameter, take six weeks to three months to resolve, and may heal with residual scarring. Herpetiform recurrent aphthous ulcers are very small (0.1 to 0.2 centimeters) and occur in groups, giving the appearance of a viral infection. However, these lesions are not viral in etiology as their name inappropriately suggests. The distribution of herpetiform recurrent aphthous ulcers is on movable mucosa. The lesions do not begin as vesicles or blisters.
The diagnosis of recurrent aphthous ulcers is usually based on the presence of characteristic historical; clinical; and, if necessary, laboratory findings. A previous history of similar lesions, the classic clinical appearance of the lesions, and the location of these lesions on freely movable mucous membranes will usually establish the diagnosis. The patient's daily medication regimen, including prescribed and over-the-counter medications, must be carefully scrutinized to ensure that the lesions are not drug related. Care should be taken to ensure that an adequate allergy and gastrointestinal history is obtained from the patient. A family history of inflammatory bowel disease may also be significant. A complete blood count with a differential as well as serum ferritin and B12 levels may be necessary for some patients.
To date, the treatment of recurrent aphthous ulcers is primarily palliative and symptomatic. Every effort should be made to eliminate predisposing allergens from the patient's diet. It is often helpful to have the patient maintain a food diary for two weeks to identify dietary triggers. Once identified, the recurrence rate will decrease commensurate with the allergen’s role in predisposing the patient to these ulcers. Therapeutic agents such as topical steroids and amlexanox 5 percent oral paste have been effective in decreasing the symptoms and healing time, but nothing has been effective in decreasing the recurrence rate unless a trigger or serum deficiency can be identified and corrected.
| Table 2: Medications Used to Treat Candidosis35 | ||||
|---|---|---|---|---|
| Medication | Forms | Dispense | Instructions | Advantages/Disadvantages |
| Ketaconazole | 200 mg tabs | 14 | Take one tablet daily with a meal until gone. | Inexpensive, high compliance, poor, absorption, must take with meal, potential for hepatotoxicity. |
| Fluconazole | 100 mg tabs | 15 | Take two tablets the first days and then one tablet daily until gone. | High compliance, good absorption, expensive, potential for hepatotoxicity. |
Topical steroids are frequently used for the treatment of minor recurrent aphthous ulcers (Table 3). These drugs seem to be effective by decreasing the inflammatory response, which results in minimizing both the symptoms and the healing time. One of the most effective topical steroid preparations is 0.05 percent fluocinonide, which is available in a cream, ointment, or gel vehicle. Patients must be warned that chronic use of topical steroids may result in mucosal atrophy as well as systemic absorption of the steroid.
 |
 |
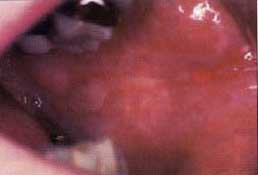
| Figure 7a. Ulcerative lichen planus in a 74-year-old female. Ulceration along the left linea alba. Note the striae peripheral to the ulceration. | Figure 7b. Appearance of the left buccal mucosa following two weeks of topical steroid therapy. |
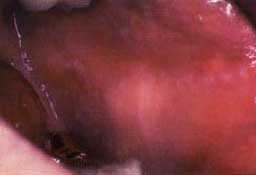
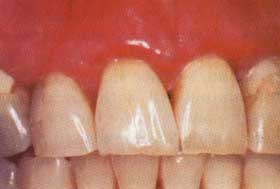
| Figure 8a. Atrophic gingival lichen planus in a 56-year-old female. Erythema of the attached gingiva, especially along the free gingival margin. | Figure 8b. Custom steroid carrier. |
 |
|
| Figure 8c. Partial resolution of the gingiva after four weeks of nightly topical steroid applications in the custom carrier. Note the overall improvement in gingival tone and the "breaking up" of the erythema of the free gingival margin. |
| Table 3: Topical Medications Used to Treat Recurrent Aphthous Ulcers35 | ||||
|---|---|---|---|---|
| Medication | Forms | Dispense | Instructions | Advantages/Disadvantages |
| Flucocinonide | 0.05% cream. | 30 gram tube. | Apply thin coat to ulcer after meals and at bedtime. Do not eat or drink for 30 min. following application. Do not use for more than two weeks. | Shortens the duration and severity of lesions, prompt relief of pain, patient must carefully follow directions, risk of mucosal atrophy absorption with prolonged use. |
| Amlexanox | 5% oral paste. | 5 gram tube. | Apply thin coat to ulcer after meals and at bedtime. Do not eat or drink for 30 min. following application. | Primary indication for use is aphthous ulcers, very safe, minimal side effects, moderately expensive. |
| Dexamethasone | elixir 0.5 mg/5ml. | 100 ml | Rinse with one teas spoonful for 3 min. four times daily, and expectorate, until lesions resolve. DO NOT SWALLOW. | Shortens the duration and severity of lesions, prompt relief of pain, patients must carefully follow directions, risk of mucosal atrophy and absorption with prolonged used, risk of secondary candidosis. |
Patients must be instructed not to use topical steroid medications on virally induced lesions. Topical steroid therapy should not exceed two weeks for any occurrence of aphthous ulcers. Patients should discontinue topical steroid use for at least two weeks before reinstituting therapy for a recurrence of lesions. Cases of disseminated recurrent aphthous ulcers can be treated with a steroid rinse such as dexamethasone elixir 0.5 mg/ 5 ml. The same information regarding steroid use should be given to the patient. The broad mucosal contact of steroid rinses may predispose the patient to secondary candidosis. In severe cases that are not responsive to topical steroids, systemic steroids such as prednisone may become necessary. When prescribing systemic steroids or steroid rinses for severe oral ulcerations, concurrent therapy with antifungal agents should be considered.
Amlexanox 5 percent oral paste is a new medication specifically indicated for the management of recurrent aphthous ulcers. 16-17 This medication has antihistamine properties and has been shown clinically to reduce the symptoms and duration of the lesions.
Other types of treatment include topical anesthetics, caustic agents, and laser ablation. Topical anesthetics such as 2 percent viscous lidocaine hydrochloride will provide temporary relief of pain. Caustic agents such as silver nitrate have been used to cauterize the central portion of the ulcer, thus providing pain relief. Cautery may result in mucosal scarring, which limits the clinical indications of this technique to cases of severe, extremely painful ulcers. Laser ablation has been used to manage aphthous ulcers in some individuals but is limited by both the availability of the laser and the cost to the patient for repeated treatments.
The most commonly occurring viral infections of the oral cavity and perioral tissues are caused by the herpes simplex virus (HSV). The herpes simplex virus has two distinct serotypes, HSV-I, which is primarily associated with oral and perioral lesions and HSV-II, which is primarily associated with genital lesions. Studies have shown that this type site predilection, however, is changing, possibly due to changing sexual practices. In a review of 336 cases of primary genital herpes, 10.7 percent were associated with HSV-I.18 A similar study of 160 cases of primary herpetic gingivostomatitis indicated that 2.5 percent of these infections were associated with HSV-I.18
Exposure to HSV-I is widespread, since HSV-I serum antibodies can be found in up to 90 percent of the American population. Primary herpetic infections occur in individuals with no previous exposure to the virus. They are seen most often, therefore, in children and adolescents. Primary herpetic infections can and do occur in adults but are often misdiagnosed by both physicians and dentists. Most often, exposure to HSV in children results in a subclinical infection. Only a small percentage of patients will develop clinical manifestations of primary herpes, most often primary herpetic gingivostomatitis, pharyngitis, or both.
The clinical manifestations of primary herpetic gingivostomatitis include systemic involvement as well as oral lesions. This disease is characterized by a rapid onset of generalized prodromal signs and symptoms, such as fever, malaise, headache, irritability and regional lymphadenopathy. The systemic symptoms are followed by the development of oral lesions.
The first intraoral manifestation is a severe generalized gingival inflammation. Within several days, this is followed by the development of oral vesicles, which can occur on any of the oral mucous membranes. The vesicles rupture very rapidly, forming shallow, ragged, extremely painful ulcerations with a yellowish center and erythematous borders. The lesions begin as numerous punctuate ulcerations that then coalesce, forming larger irregular ulcerations. Extraoral lesions occurring on the lips will often appear crusted. The oral lesions a usually accompanied by severe pain foul odor, and increased salivation.
Acute primary herpetic infection may also present as pharyngitis without any other oral lesions. This occurs most often in adolescents and is characterized by a sore throat, dysphagia, cervical lymphadenopathy, fever, and headache.
The diagnosis of primary herpetic gingivostomatitis is most often based on the presence of prodromal systemic signs and symptoms in addition to characteristic clinical appearance generalized oral ulcerations. Diagnostic tests such as viral isolation in tissue culture and direct immunofluorescent antibody testing can be used to confirm the diagnosis, but they are not routinely used in an otherwise healthy patient.
Acute primary herpetic oral infections can vary considerably in severity and duration. In a healthy individual the disease is self-limiting and lasts proximately 10-14 days; and the lesions heal without scarring. Since is an acute viral infection, the usual supportive measures such as the maintenance of adequate fluid intake nutrition should be undertaken to prevent dehydration and electrolyte balance. Patients often present with severe pain. Systemic analgesics such as acetaminophen with or without codeine are useful for the control pain. Aspirin and nonsteroidal antiinflammatory medications should avoided in acute viral infections.
Systemic antiviral agents such acyclovir are not routinely indicated for primary herpetic gingivostomatitis except in severely immunocompromised patients or when there is ophthalmologic involvement. However, if the of acyclovir is considered, it will be most effective if prescribed very early in the clinical course of the infection. The usual dosage of acyclovir is 200 mg, five times daily for seven to 10 days (Table 4)19. The current Food and Drug Administration recommendation is that this regimen be used to treat oral herpes only in immunocompromised patients.
| Table 4: Medications Used to Treat Herpetic Infections35 | ||||
|---|---|---|---|---|
| Medication | Forms | Dispense | Instructions | Advantages/Disadvantages |
| Penciclovir | 1 % cream. | 2 gram tube. | Apply every two hours during working for four days. Treatment should be started during the prodromal symptoms, if possible. | High compliance, decreased duration and pain of episode, primary indication is RHL, safe expensive. |
| Acyclovir* | 200 mg capsules | 50 | Take one capsule five times daily until gone. | Safe, inexpensive, must be initiated during prodrome to be effective. |
| Acyclovir** | 400 mg capsules. | 14 | Take two capsules daily starting one day prior to anticipated sun exposure until gone. | Safe, inexpensive. |
| * Recommended regimen, for primary herpetic gingivostomatitis. The current Food and Drug Administration recommendation is that this regimen be used to treat oral herpes only in immunocompromised patients. ** Recommended prophylactic regimen for severe episodes of recurrent herpes labialis in a patient at risk for sun exposure. The current Food and Drug Administration recommendation is that this regimen be used to treat oral herpes only in immunocompromised patients. |
||||
Approximately 30 percent to 40 percent of patients who have been exposed to HSV will develop recurrent infections that can present as either recurrent herpes labialis or recurrent intraoral herpes. These lesions represent reactivation and not reinfection of the HSV, which persists in a latent state in the trigeminal ganglion. The lesions are characterized by a mild clinical course and can be triggered by exposure to sunlight, fatigue, stress, hormonal changes such as menstruation, gastrointestinal disturbances, and oral trauma.
Recurrent herpes labialis (fever blister, cold sore) is preceded by prodromal signs or symptoms such as burning, tingling, tautness, soreness, or swelling in the site where the lesions will develop. Within a short time, small vesicles develop in clusters along the vermilion border of the lips (Figure 5). The vesicles rupture, resulting in ulcers that can coalesce to form larger irregular ulcerations with a crusted surface. In a healthy individual, lesions heal in seven to 14 days without scarring.
The diagnosis of recurrent herpes labialis is based on its characteristic clinical appearance. The treatment is still primarily symptomatic. Ice, ether, chloroform, and rubbing alcohol have been used as topical agents with mixed results. Historically, topical antiviral agents such as acyclovir ointment have also been used with limited success primarily because of poor cutaneous absorption. The Food and Drug Administration has recently approved penciclovir 1 percent topical cream for the treatment of recurrent herpes labialis (Table 4).20 This cream may reduce the severity and duration of the viral outbreak.
Lysine tablets have also been used with varying degrees of success. Some investigators reported that lysine resulted in milder episodes of herpes labialis if taken in high doses (2-3 gm) at the first prodromal signs.21 In some individuals, it was also found to be effective in preventing recurrences if the patient continued to take 1,000 mg per day.
Systemic antiviral agents, such as acyclovir, are primarily used in severe cases of mucocutaneous or ophthalmologic herpes simplex infections in immunocompromised patients. However, in patients known to have recurrent herpes labialis induced by exposure to sunlight, a prophylactic regimen of acyclovir 400 mg twice daily may be employed if prolonged actinic exposure is anticipated (Table 4). 22
In healthy patients, recurrent intraoral herpes occurs less frequently than herpes labialis. Intraoral herpes may be triggered by dental therapy or trauma from mastication. The intraoral lesions rupture very rapidly, leaving small punctuate ulcerations. These ulcers can coalesce as the lesion progresses, forming larger irregular ulcerations. The lesions occur primarily on oral tissues that are firmly bound to the underlying bone, i.e., the hard palate, attached gingiva, and alveolar ridges.
The diagnosis of these lesions is based on the clinical appearance of the lesions and their location. The amount of discomfort associated with these lesions varies, and they heal without scarring in seven to 14 days. The management of pain in recurrent intraoral herpes would involve the same topical palliative agents as were recommended for the painful lesions of primary herpetic gingivostomatitis.
Patients must be informed of the infectious nature of both acute and recurrent oral herpetic conditions. The vesicles of oral herpetic infections are extremely contagious, so care must be exercised to avoid autoinoculation of other mucosal sites as well as transmission to others. All stages of these viral lesions are potentially infectious until complete re-epiethelialization has occurred.
| Table 5: Medications Used to Treat Lichen Planus35 | ||||
|---|---|---|---|---|
| Medication | Forms | Dispense | Instructions | Advantages/Disadvantages |
| Flucocinonide | 0.05% cream ointment gel. | 30 gram tube. | Apply thin coat to ulcer after meals and at bedtime. Do not eat or drink for 30 min. following application. DO NOT USE FOR MORE THAN TWO WEEKS. | Shortens the duration and severity of lesions, for relief of pain, patient must carefully follow directions, risk of mucosal atrophy absorption with prolonged use; gel form should be used with custom carrier(s) |
| Clobetasol propionate | 0.05% ointment. | 30 gram tube. | Apply thin coat to ulcer after meals and at bedtime. Do not eat or drink for 30 min. following application. DO NOT USE FOR MORE THAN TWO WEEKS. | Shortens the duration and severity of lesions, for relief of pain, patient must carefully follow directions, risk of mucosal atrophy and absorption with prolonged use; risk of systemic absorption. |
| Dexamethasone | elixir 0.5 mg/5ml. | 100 ml | Rinse with one tea spoonful for 3 min. four times daily, and expectorate, until lesions resolve. DO NOT SWALLOW. | Shortens the duration and severity of lesions, prompt relief of pain, patients must carefully follow directions, risk of mucosal atrophy and absorption with prolonged used, risk of secondary candidosis. |
Lichen planus, a disorder of un known etiology, represents the most common dermatologic disease with oral manifestations. The skin lesion occur as violaceous papules with a fine scale on the flexor surface of the arms and legs. The oral lesions vary greatly in appearance and frequently represent the only clinical sign of disease. Lichen planus is usually found in patients older than 40 and is frequently associated with stress and anxiety.23-24 Numerous medications have been reported to cause oral lichenoid drug reactions. Commonly encountered medications may include thiazide diuretics and tetracyclines. Dental amalgam restorations with direct mucosal contact have also been implicated in lichenoid reactions. The patient may be unaware of the intraoral form of the disease because it is often asymptomatic. Therefore, the oral soft tissues of patients with signs and symptoms of dermal lichen planus must be examined.
Oral lichen planus is often identified by the presence of fine reticular white lines (striae of Wickham) on the lateral borders of the tongue, buccal mucosa, and gingiva (Figure 6). However, striae are not always present, especially in the ulcerative form of the disease; therefore, definitive diagnosis is made by biopsy. Although prospective studies have failed to demonstrate that lichen planus is a premalignant disorder, it is recommended that all patients exhibiting this condition intraorally, particularly those who have had the ulcerative form, receive long-term follow-up (Figure 7).26-28
Fluocinonide cream, ointment, or gel is effective in treating mucosal lesions and has not been shown to cause adrenal suppression (Table 5).29 Ultrapotent topical steroids such as clobetasol propionate ointment may also be used intraorally and appear to be safe and effective. However, because of potential systemic adverse reactions from long-term use of ultrapotency topical steroids on the skin, patients must be monitored regularly.31
Occlusive steroid therapy using custom-made flexible mouth guards to localize fluocinonide gel is extremely effective in controlling the gingival lesions of lichen planus.32-33 Fluocinonide 0.05 percent gel is applied to the mouth guards, and patients are instructed to wear them for 30 minutes per application. Therapy can be instituted with a regimen of four applications daily, preferably after meals and at bedtime. The frequency of daily use depends on the response of the lesions. Long-term control of persistent lesions can often be achieved with a 30-minute application every other evening.
Disseminated lesions can be controlled with dexamethasone rinses. Refractory, persistent, localized lesions respond very favorably to intralesional injection of triamcinalone (10 mg/ml) in lidocaine. Secondary candidosis should be considered when ulcerated lesions remain refractory to conventional therapy or when an unexplained relapse occurs in a patient using topical steroids.34 Systemic corticosteroids will resolve oral lesions that occur coincidently with skin lesions of lichen planus, but recurrence of oral lichen planus is likely. Use of systemic steroids for longer than two weeks at doses greater than 40 mg daily is likely to result in side effects that may not resolve once the steroids are discontinued. These side effects include oral candidosis, hypertension, fluid retention, gastrointestinal discomfort, insomnia, fat deposition, and skin rash. Intraoral mucosal conditions that are refractory to therapeutic trial or remain present for longer than two to four weeks should be biopsied to establish a definitive diagnosis.
Management of painful oral mucosal conditions may be either topical or systemic. Oral therapy should address patient nutrition and hydration, oral discomfort, oral hygiene, and management of secondary infection, as well as local control of the disease process. Depending on the extent, severity and location of oral lesions, consideration should be given to obtaining a consultation from a dentist who specializes in oral medicine, oral pathology, oral surgery, or periodontics. If the oral lesions are painful enough to limit normal dietary intake, nutritional supplementation and adequate hydration must be stressed to the patient. Commercially available weight-control beverages serve this purpose in a cost-effective manner. Cold beverages or ice chips may provide temporary relief of oral pain. Citrus fruits, carbonated beverages, and other acid-containing or spicy foods will exacerbate oral discomfort, so they should be avoided.
| Table 6: Medications Used to Treat Lichen Planus35 | ||||
|---|---|---|---|---|
| Medication | Forms | Dispense | Instructions | Advantages/Disadvantages |
| Lidocaine HCI | 2% viscous. | 100 ml bottle. | Apply with cotton swap or rinse for two minutes prior to meals and expectorate. DO NOT GARGLE OR SWALLOW. | Safe, inexpensive, fast-acting pain relief, short-duration, may compromise gag reflex. |
| Dyclonine HCI | 0.5% or 1.0%. | 1 ounce bottle. | Apply with cotton for two minutes prior to meals and expectorate. DO NOT GARGLE OR SWALLOW. | Safe, inexpensive, fast-acting pain relief, long-duration, may compromise gag reflex. |
| Diphenhydramine HCI and magnesium hydroxide/ aluminum hydroxide | children's elixir suspension. | OTC Mix equal (50% mixture of each) | Rinse with two minutes as necessary for pain and expectorate. | Safe, inexpensive, fast-acting pain relief, provides coating over ulceration, short duration, children's elixir contains no alcohol. |
| Sucralfate | suspension. | 14 ounce bottle | Rinse with two teaspoonful two minutes prior to meals and expectorate. | Safe, fast-acting pain relief, provides coating over ulceration, moderate duration, expensive. |
Symptomatic relief can be provided with topical preparations such as 2 percent viscous lidocaine hydrochloride, or 0.5 percent dyclonine hydrochloride applied prior to meals to facilitate eating (Table 6). Topical anesthetics can be used as a rinse in adults but should be applied with a cotton swab in a child so that the child does not swallow the medication. Swallowing these anesthetics is contraindicated, in part because they may interfere with the patient gag reflex. Symptomatic relief can also be obtained by mixing equal parts of diphenhydramine hydrochloride elixir and magnesium hydroxide/aluminum hydroxide. Children's formula diphenhydramine hydrochloride elixir does not contain alcohol. Sucralfate suspension may also be used prior to meals. The diphenhydramine mixture an the sucralfate coat the ulcerated lesions and allow the patient to eat more comfortably.
Mouth rinses containing a hydroalcoholic vehicle should be avoided because of the oral discomfort that will result from their use. The amount of oral discomfort experienced by patients with oral mucosal lesions varies and can often be controlled without narcotic analgesics.
Meticulous oral hygiene is mandatory for these patients. Mucosal lesions contacting bacterial plaque present on the dentition are more likely to become secondarily infected. Patients should be seen by the dentist or hygienist for scaling and root planning, under local anesthesia when necessary, in all cases where oral hygiene is suboptimal. Patients must be encouraged to brush and floss their teeth after meals in a gentle, yet efficient manner. This may be enhanced by placing a soft toothbrush under hot water to further soften the bristles. Tartar-control toothpastes containing calcium pyrophosphate should be avoided because of their caustic nature and reported involvement in circumoral dermatitis.
Secondary infection of ulcerative oral mucosal lesions is most commonly fungal in etiology. While long-standing oral ulceration alone may predispose some patients to secondary candidosis, therapeutic regimens of antibiotics or steroids will increase the frequency of mycotic infection in susceptible individuals. Bacterial infection is less common. Unless the lesions become indurated or purulent or are associated with an acute tender lymphadenopathy, antibiotic coverage should not be necessary.
Michael A. Siegel, DDS, MS, is an associate professor in the Department of Oral Medicine and Diagnostic Sciences at the University of Maryland at Baltimore Dental School.
Copyright CDA Journal. Vol 27. No. 8. August 1999
Reprinted with permission.