
| Contents | Previous | Next |
by Mark L. Bernstein, DDS
Abstract: Chronic lichenoid or leukoplakic oral mucosal lesions are a common cause of morbidity and concern. Many of these are reactive or inflammatory lesions; but they can also represent other disorders, including dysplasia. These lesions are caused by a variety of irritants and allergens such as systemic drugs, dental restorations and prostheses, oral health care products, foods, habits, and candida. Indiscriminate use of steroids to treat these lesions empirically is contraindicated; management should be aimed at discovery and removal of the cause. The proposed sequence of investigation is intended initially to eliminate any obvious etiology and, if that is unsuccessful, to rule out candida or determine a histologically diagnosable disease. If a biopsy shows nonspecific or lichenoid mucositis and the lesions are symptomatic, the investigation is directed to the more obscure and speculative causes that require expensive and time-consuming trial-and-error elimination of various agents.
The practicing clinical oral pathologist is often referred patients with oral lesions that present diagnostic and management challenges. Among the most frequent of these is a group referred to as “chronic nonspecific mucosal lesions.” These disorders represent low-grade physical or chemical injuries or delayed hypersensitivity reactions that share a common clinical presentation as persistent, nonspecific focal white or mixed red and white lesions. These conditions can cause considerable morbidity; and they defy diagnosis and resist therapy, making them a chronic problem for the patients who live with them and the dentists who treat them.
Chronic nonspecific mucosal lesions first come to a dentist’s attention during a routine oral exam or in response to a patient complaint of roughness, a burning sensation, or pain. Clinically, they appear as localized or multifocal white or mixed red and white (speckled) lesions that range from flat to plaque like and may be granular and partly erythematous. Such lesions are nondescript and not recognizable as a specific disease, thus meriting the clinical term leukoplakia or erythroleukoplakia. Others with striations, ulceration, or atrophy resemble the many forms of lichen planus and are referred to as lichenoid. The designation “lichenoid” is also a microscopic term describing the bandlike subepithelial lymphocytic infiltrate typical of lichen planus but also seen in other conditions.
A clinician will be compelled to investigate these lesions because of their chronicity, symptomatology, or perceived risk of malignancy. What often happens next is that the uncertainty of diagnosis will prompt a biopsy to provide a solution. Regretfully, the histopathology of reactive or allergic lesions is often nonspecific, showing only chronic inflammation with epithelial changes of hyperkeratosis, atrophy, hyperplasia, and superficial erosion. A microscopic diagnosis of nonspecific inflammation or lichenoid mucositis is then made. Although this diagnosis rules out malignancy, it does little to direct treatment. Consequently, the lesion persists, and the patient is relegated to symptomatic treatment that offers only temporary relief. This chronicle of events is unfortunate because many of these lesions have a specific cause that if approached properly can be identified and removed to effect a permanent cure. It is the objective of this article to call attention to some recognized etiologic agents and correlate them with subtle lesional patterns that suggest a precise diagnosis that can be pursued through additional history and testing.
When any clinical white or speckled lesion is encountered, most knowledgeable clinicians will take a thorough history, asking about topical irritants; habits, including tobacco and alcohol use; medical conditions; and presence of skin lesions. Additionally, they will look for obvious local factors conducive to lesion formation. This information, combined with lesional patterns and locations, will allow a tenable clinical diagnosis of such distinctive conditions as nicotine stomatitis, snuff dipper’s keratosis, leukoedema, cheek biting, geographic tongue, and classic lichen planus. The focus of this article is the remaining wastebasket of clinically and histologically undefined lesions for which a specific cause cannot be ascribed.
| Table 1. Causes of Chronic Nonspecific White and Speckled Red-and White Mucosal Lesions |
|---|
|
Table 1 lists the causes of clinically nondescript white and speckled lesions. One might wonder, if these lesions have determinate etiologies, why they cannot be more easily characterized. The reason a causative agent cannot be attributed to a particular lesion is eightfold:
Over the years, investigators have extracted from the wastebasket of lichenoid and leukoplakic lesions several distinct entities that - by virtue of subtle clinical, histologic, and historic data - facilitate a more precise diagnosis.
 |
  |
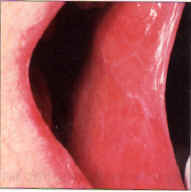
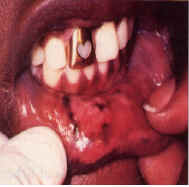
| Figure 1. Diffuse network of white striae in buccal mucosa bilaterally of a woman treated for hypertension with methlydopa. | Figure 2a. and b. Localized and pigmented lesions in intimate contact with a shell crown. The lesions, present several months, were associated with swelling and pain. Histology showed epithelial thickening, melanosis and severe chronic inflammation with eosinophils–characteristic of an allergic response. Melanin pigmentation often accompanies chronic ulcerinflammatory lesions. |
 |
  |
| Figure 3. Leukoplakia confined to an area of contact with a large amalgam restoration proximate to gold. In this case, galvanism might contribute by accelerating corrosion. Replacement of the amalgam filling is indicated. | Figure 4a. and b. Erythroleukoplakia of the lateral tongue. Improvement of the erythroleukoplakia shown in 4a following replacement of both the amalgam filing in No. 30 and the gold crown in No. 31 with two porcelain-surface crowns. |
Certain medications are known to cause generalized or localized oral lesions that can be indistinguishable from lichen planus clinically and microscopically (Figure 1). The medications are thought to alter the antigenicity of epithelial cells, rendering them targets for sensitized T-lymphocytes. In effect, the reactions can be thought of as lichen planus induced by drugs. The essential difference is that LDE remits when the drug is withdrawn.
Table 2 lists groups of medications associated with oral lichenoid lesions. In practice, any drug taken by a patient should be investigated. Lichenoid drug eruptions show a latent period averaging one year, with a full range of one month to three years, before development of lesions. The latent period depends on the drug, its dosage, drug interactions, and a patient’s individual susceptibility. In all patients diagnosed with symptomatic oral lichen planus, suspect medications antedating the lesions should be discontinued or substituted, if possible, after consultation with the prescribing doctor. It might require from two weeks to two years for lesions to disappear. Resolution can be accelerated with topical steroids. It must also be realized that both lichen planus and the use of drugs producing LDE are common in older adults, but LDE itself is infrequent. Thus, in many cases the medication is unrelated - a fact that may require months to determine when the lesions fail to remit after cessation of the drug.
Lichenoid, leukoplakic, or erythroleukoplakic lesions are occasionally noted adjacent to dental restorations or prosthetic appliances. They are typically located unilaterally on the buccal mucosa, lateral tongue, or gingiva where they are localized to an area in contact with the material or may extend beyond the contact area (Figures 2 a and b). There are often symptoms of discomfort of burning.
The clinical differentiation between idiopathic lichen planus or dysplasia and a lichenoid lesion induced by a restoration cannot be made with confidence. Restorative dentistry is prevalent in older adults where its coexistence with lichen planus or dysplasia might be fortuitous. Since lichen planus and dysplasia are clearly more common than a contact reaction, replacement of restorations based on a mere concordance of lesion and dental material is impractical. Yet, a growing number of studies indicate that lichenoid reactions to dental material do occur and that lesions resolve following removal of the contacting agent. How then does one determine when removal of dental work is justifiable?
In a study of 275 patients having skin or oral allergic lesions, the most frequent sensitizers were mercury (53 percent); followed by chromium, nickel, cobalt, and tin (35 percent to 32 percent); and platinum, iridium, palladium, cadmium, zinc, molybdenum, and gold (19 percent to 13 percent). Amalgam – with its content of silver, mercury, tin, copper, and traces of palladium and zinc – is the most likely offender (Figure 3).
Partial denture frameworks contain chromium and cobalt, and cast crowns may contain, in addition to gold and platinum, nickel, palladium, iridium, cadmium, and molybdenum. Even high noble alloys can include trace metals and impurities. Acrylic (denture baseplates and retainers), nickel (orthodontic wire), composite restorations, and BISGMA have been cited as mucosal irritants or allergens. Typically, the metal salts rather than the elemental metal incite the reaction, which is why the inert nonionizing metals are less likely to induce sensitivity.
The experience of acute galvanic shock is familiar to patients when new amalgam fillings contact old restorations or metal foil. The shock sensation is due to a transfer of electrons between metals of different electromotive potential in an electrolyte (saliva). Chronic, asymptomatic low-grade galvanism, occurring where dissimilar metals contact each other, has been implicated as a cause of lichenoid lesions, leukoplakia, and oral cancer; but proof is lacking. Oral electric currents cannot be recorded easily, and it is difficult to separate the influences of galvanism from primary metal reactions. Galvanism may contribute to lesion development by accelerating the rate of corrosion that forms the metal salts responsible for hypersensitivity (Figure 3).
| Table 2. Drugs Commonly Associated With Lichenoid Reactions |
|---|
|
The decision to remove or replace a dental material adjacent to a lesion must include an assessment of the risk-to-benefit ratio, factoring in the likelihood of causation, cost, complexity, symptoms, patient stress, and concern about malignancy. Removable prostheses or isolated corroded/defective restorations are more conducive to removal than fixed bridgework. Lesions confined to the area of contact are more likely to respond to elimination of a material than lesions that extend beyond the confines of the material. If the composition of the material is known, each component can be evaluated with patch testing. Two percent to 5 percent of the general population is allergic to mercury, while 16 percent to 62 percent of individuals with adjacent lichenoid lesions are found to be allergic to the metal.
Test results must be interpreted with caution. Dermal allergy testing can give false negative results if the oral lesion represents an irritation by the restoration rather than a hypersensitivity. False positive results can also occur for two reasons: skin is more sensitive than mucosa; and metal salts are usually used in patch tests, which is irrelevant if the oral metal does not ionize in vivo. When the constituents of a restorative material are not known, they can often be determined by name brand. If the brand name is not known, a small sample can be recovered with a handpiece and submitted for elemental analysis using energy dispersive X-ray microanalysis. This service is available from institutions that have a scanning electron microscope. Such sophisticated techniques are advocated for patients with extensive fixed dental appliances.
 |
 |
| Figure 5a and b. "Kissing" granular leukoplakias of the buccal mucosa and tongue in a 54-year-old cinnamon-gum chewer. | |
 |
 |
| Figure 5c and d. Full resolution of the case in 5a and b within two weeks of discontinuance of the cinnamon-gum chewing. | |
 |
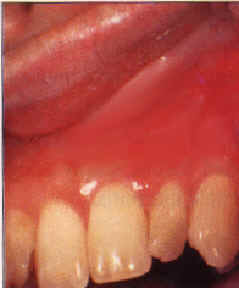 |
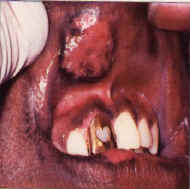
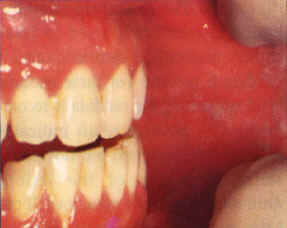
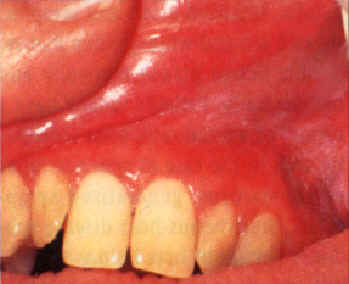
| Figure 6a and b. Asymptomatic, irregular, extensive leukoplakia of the anterior maxillary vestibule, mucolabial fold and alveolar mucosa extending from the right canine to the left molar. The histology showed mild dysplasia. The patient, a 54-year-old woman, used Viadent mouthwash and toothpaste for seven years. Partial improvement after 10 months. | |
 |
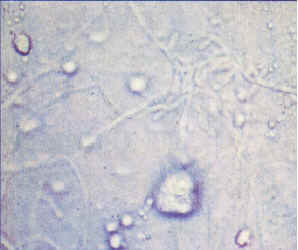 |
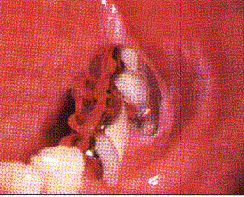
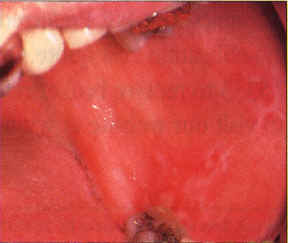
| Figure 7. Red-and-white lichenoid lesion with cytologic evidence of candida. Complete resolution followed antifungal therapy. | Figure 8. Unstained candidal pseudohyphae and spores as seen in a KOH prep, original magnification, 400x |
 |
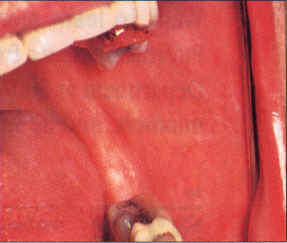 |
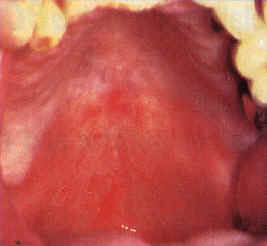
| Figure 9a and b. Lichen planus in a 66-year-old woman with no contributory contacting materials or medications. Following a negative smear for candida, Temovate and Orabase B was applied topically for a period of three weeks. Resolution was complete. | |
Upon removal of a causative material, resolution of the lesion can be expected in one to 12 months with relief of symptoms within three months (Figures 4a and b). However, this is too long a wait if the lesion is located in a high-risk area for oral cancer such as the lateral tongue, in which case a biopsy is recommended before replacement of the dental restoration.
Toothpastes and mouthwashes contain a multitude of agents recognized as allergens. Additionally, ingredients such as pyrophosphate and zinc citrate (tartar control) and sodium lauryl sulfate (surfactant) are contact irritants that have been implicated in mucosal sloughing and aphthaeform ulcers, respectively. Up to 2 percent of toothpaste users report problems.
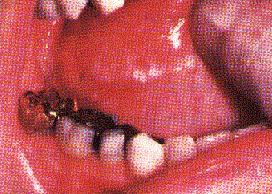
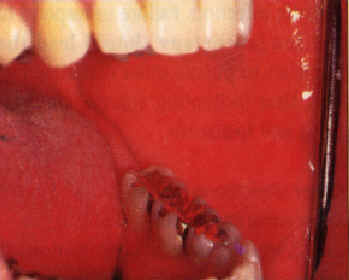
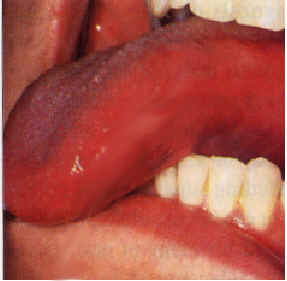
The most notorious sensitizing agent in food and oral health care products is cinnamon flavoring, which causes contact mucositis in an untold number of patients. The lesions vary in appearance and location, corresponding to patient habits. Cinnamon- flavored chewing gum is most often implicated, and lesions form adjacent to the contact area, along the occlusal line of the buccal mucosa with or without corresponding “kissing” lateral tongue lesions (Figures 5a and b). They may be unilateral or bilateral. Some are lichenoid, whereas others are eroded and granular.
Clinically, they may mimic lichen planus, cheek biting, snuff dipper’s keratosis, candidiasis, hairy leukoplakia, or dysplasia. A directed inquiry of patient habits may identify the offending agent; but too often, patients are unaware of cinnamon use, not realizing that the brand of gum they chew is cinnamon-flavored or not knowing that foods they ingest contain cinnamon (e.g., sweet vermouth, chili). Often, the gum habit is a recently instituted substitute for smoking. Other vehicles such as candy or even dental floss might deliver cinnamon, resulting in different lesional patterns. The lesions may cause months of discomfort, burning, pain, or a rough feeling. When biopsied, a characteristic histologic finding virtually identifies the lesion as a cinnamon reaction.
These lesions are characterized by a pronounced perivascular chronic inflammatory infiltrate below a lichenoid surface change. Based on the predictive histology alone, patients have been contacted following biopsy and “accused” of a cinnamon habit. The patients are incredulous but grateful when the lesions remit within two weeks of cinnamon cessation (Figures 5c and d).
Damm reported on 88 patients who developed a peculiar white mucosal patch extending across the maxillary labial mucosa, vestibule, and alveolar mucosa. Histologically, these lesions showed hyperkeratosis, chronic mucositis, and mild dysplasia. These lesions were further investigated because of the unusual location of the dysplasia, which did not correspond to that seen with tobacco use. Upon further history, nearly 85 percent of these patients used sanguinaria-containing Viadent mouthwash and/or toothpaste for varying amounts of time.
Once formed, these kinds of lesions are slow to resolve (Figures 6a and b). The histologic finding of dysplasia is worrisome, although sanguinaria-related lesions do not show a preneoplastic profile biochemically, according to Eversole and colleagues. The clinical lesion is so unusual that it should prompt questioning about the patient’s dentifrice and mouthwash habits.
Ironically, lichen planus is included in the differential diagnosis of lichenoid lesions. Lichen planus is an idiopathic, chronic, recalcitrant disorder of the skin and mucous membranes thought to represent a Type IV hypersensitivity to an exogenous agent that alters keratinocytes. It is mediated by activated cytotoxic T-lymphocytes that damage basal keratinocytes and the basement membrane. Up to 2 percent of the population is affected, particularly women older than 40.
When lichen planus shows classic white striae arborizing throughout the buccal mucosa bilaterally in a patient who is not taking medications known to cause lichenoid reactions, the diagnosis is straightforward. But many cases are atypical. Atrophic or erosive forms present as fissured white plaques on the dorsal tongue, as a desquamative gingivitis, or as erythroleukoplakia with or without well-delineated yellow-tan ulcers. These are difficult to diagnose clinically, and so reliance is placed on a biopsy.
On the skin, the application of fastidious histologic criteria can establish the diagnosis of idiopathic lichen planus in 90 percent of cases. Oral lichen planus does not necessarily exhibit these pathognomonic features. The microscopic features of oral lichen planus may be altered because of factors including superimposed inflammation or candidiasis, attempts at therapy, varied histology of oral tissues, and poor selection of biopsy site. Biopsies from the gingiva and areas of ulceration are frequently non-diagnostic. Neither histology nor clinical appearance are dependable as a gold standard to firmly establish the diagnosis of lichen planus in the oral mucosa. Consequently, idiopathic oral lichen planus may fall into the group of undistinguished lesions simply designated as lichenoid mucositis.
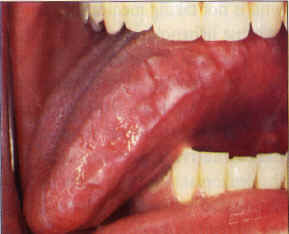
The consideration of candidiasis in the differential diagnosis of white and speckled lesions is relevant for three reasons. First, chronic atrophic or hyperplastic candidiasis may present as a lichenoid lesion (Figure 7). Second, candida may be superimposed on other lesions in this group and alter the lesional appearance and confound diagnosis. Third, steroid therapy, which is commonly used to treat chronic mucositis, is contraindicated if candida is present. For these reasons, cytologic testing for candida is recommended. A positive result indicates that candida is present but not necessarily the cause of the lesion. Antifungals should be prescribed. If the lesion completely resolves, it can be attributed to candida; and the patient should be evaluated for local and systemic factors that favor this opportunistic infection. Failure to respond to antifungal therapy should lead the clinician to pursue other causative factors, thus facilitating appropriate diagnosis and treatment.
There are several uncommon or rare causes of localized lichenoid lesions that are included for completeness. Most can be ruled out by history. Lupus erythematosus will produce painful, localized ulcers and atrophic, striated red-and-white lesions that resemble lichen planus clinically and histologically. Patients usually have a history of a facial rash; solar sensitivity; and, in the case of systemic lupus erythematosus, generalized symptoms. Graft-versus-host disease produces lichenoid oral lesions. This is not surprising because graft lymphocytes reacting against antigenically dissimilar host epithelium simulates the pathogenesis of lichen planus. A history of a bone marrow allograft establishes the diagnosis.
Chronic ulcerative stomatitis is a rare and recently recognized disease that is responsible for widespread oral ulcer; and desquamative gingivitis mimicking lichen planus clinically and histologically. It is most frequent in women older than 40. If it is considered in the differential diagnosis, immunofluorescent findings are diagnostic. Direct and indirect immunofluorescence is strongly positive for stratified epithelium-specific antinuclear antibody. The direct method also shows fluorescence of epithelial nuclei with anti-IgG. The disease is resistant to steroids but responds to therapy with hydroxychloroquine (Plaquenil), 200 mg b.i.d.
Chronic nonspecific reactive mucosal lesions are caused by so many disparate agents that no single management protocol can be applied to all cases. However, it is useful to be guided by a decision tree that reaches an appropriate end point expediently without placing the patient at unnecessary medical or financial risk. A logical progression should include:
A patient history and oral examination are the most immediate sources of diagnostic information and supply the foundation for further decision-making:
It is unwise to treat an undiagnosed lesion with a trial of steroids for several reasons:
Ruling out candida involves a simple office screening test that should be routinely performed with white, red, and speckled lesions. If the clinician owns a microscope, a potassium hydroxide (KOH) prep can be examined within minutes. A smear of the lesional surface is applied to a slide onto which is placed a drop of aqueous 10 percent KOH. The slide is coverslipped, gently heated over a Bunsen burner to hydrolyse the epithelial cells and examined microscopically for evidence of yeast (Figure 8). If a microscope is not available, or if there is not a visible amount of material on the slide, the smear should be sprayed with a cytology fixative and sent to an oral pathology laboratory with instructions to evaluate for candidiasis using a periodic acid Schiff (PAS) stain.
After the elimination of candida and any easily correctable causes of the lesion, it is time to consider more speculative etiologies such as medications and contact reactions. Some of these reactions require several months to resolve, even if the cause is eliminated. This is an unacceptable waiting period if a histologic diagnosis has not been determined. If a biopsy has not yet been performed, it is now indicated.
If a diagnosis of nonspecific or lichenoid mucositis is returned and the lesion is symptomatic, it is appropriate to eliminate suspect drugs, oral health care products, foods, and dental restorations and materials. This may require the support of allergy testing, as deemed necessary and within reason. Ultimately, removal of extensive serviceable restorations or discontinuance of a necessary medication may exceed the risk- to-benefit ratio and is simply not acceptable, particularly if the causation is in doubt.
This is one area in the management of mucosal lesions where the interplay of options calls for judgment. Some decisions, like ruling out malignancy in a high-risk location, are compelling. Others, such as replacing a medication or costly dental work, are discretionary and largely the decision of the patient after a full disclosure has been made and informed consent obtained.
If a symptomatic, biopsy-proven chronic inflammatory lesion persists despite a comprehensive diagnostic workup and reasonable elimination of suspect reactants, either the cause was not discovered, the lesion is idiopathic, or it has not been given adequate time to resolve. Topical steroids may be used if there are no medical contraindications. Triamcinolone acetonide 0.1 percent in Orabase B is a low-strength steroid preparation.
Higher potency steroids such as fluocinonide 0.05 percent or clobetasol 0.05 percent gel mixed 1:1 with Orabase B can be used as well. A small amount is smeared across the lesion, t.i.d., after meals and before bedtime for two to three weeks and then as needed if symptoms recur (Figures 9a and b).
Systemic steroids are reserved for extensive, debilitating lesions that do not respond to topical application. After an initial burst of 30 to 45 mg of prednisone, the dose is adjusted to the lowest amount needed to prevent recurrence. For long-term treatment, alternate day therapy is useful; and adjunctive treatment with agents such as azathioprine or levamisole can be added to decrease side effects. If at any time during the course of seemingly successful steroid treatment symptoms worsen, the patient should be clinically re-evaluated and checked for candida. Treatment with steroids and immunomodulating agents may require consultation with the patient’s physician.
Mark L. Bernstein, DDS, is a professor of oral and maxillofacial pathology in the Department of Surgical and Hospital Dentistry at the University of Louisville School of Dentistry.
Alanko K, Kanerva L et al, Oral mucosal diseases investigated by patch testing with a dental screening series. Contact Dermatitis 34:263-7, 1996.
Damn DD, Curran A, et al, Leukoplakia of the maxillary vestibule - an association with Viadent? Oral Surg Oral Med Oral Pathol 87(1):61-6, 1999.
DeRossi SS and Greenberg MS, Intraoral contact allergy: a literature review and case reports. JADA 129(12):1435-41, 1998.
Elder D, Elenitsas R et al, Lever’s Histopathology of the Skin, 8th ed. Lippincott Raven, Philadelphia, 1997.
Eversole LR, Eversole GM and Kapcik J, Oral sanguinaria associated keratosis: a comparative study with other oral keratotic and dysplastic lesions. Abstract No. 6, 52nd AAOMP Annual Meeting, Dallas, 1998
Halevy S and Shai A, Lichenoid drug eruptions. J Am Acad Dermatol 29:249-55, 1993.
Koch P and Bahmer FA, Oral lichenoid lesions, mercury hypersensitivity and combined hypersensitivity to mercury and other metals: histologically-proven reproduction of the reaction by patch testing with metal salts. Contact Dermatitis 33:323-8, 1995.
Koch P and Baum HP, Contact stomatitis due to palladium and platinum in dental alloys. Contact Dermatitis 34(4):253-7, 1996.
Korstanje MJ, Drug-induced mouth disorders. Clin and Exp Dermatol 20(1):10-8, 1995.
Lewis JE, Beutner EH, et al, Chronic ulcerative stomatitis with stratified epithelium-specific antinuclear antibodies. Int J Dermatol 35(4):272-5, 1996.
Lozada-Nur F and Miranda C, Oral lichen planus: topical and systemic therapy. Seminars Cutaneous Med Surg 16(4):295-300, 1997.
Lu SY, Chen WJ, Eng HL, Dramatic response to levamisole and low-dose prednisolone in 23 patients with oral lichen planus : a 6-year prospective follow-up study. Oral Surg Oral Med Oral Pathol 80(6):705-9, 1995.
McCarten BE and McCreary CE, Oral lichenoid drug eruptions. Oral Disease 3(2):58-63, 1997.
Miller RL, Gould AR, Bernstein ML, Cinnamon-induced stomatitis venenata: clinical and characteristic histopathologic features. Oral Surg Oral Med Oral Pathol 73:708-16, 1992.
Neville BW, Damm DD, et al, Oral and Maxillofacial Pathology. WB Saunders Co, Philadelphia, 1995.
Ostman PO, Anneroth G, Skoglund A, Amalgam-associated oral lichenoid reactions: clinical and histologic changes after removal of amalgam fillings. Oral Surg Oral Med Pathol 81(4):459-65, 1996.
Pang BK, Freeman S, Oral lichenoid lesions caused by allergy to mercury in amalgam fillings. Contact Dermatitis 33:423-7, 1995.
Rick GM, Case 2 - Lichenoid mucositis consistent with contact mucositis. J Cal Dent Assoc 25(8):545-52, 1997.
Sainio EL, Kanerva L. Contact allergens in toothpaste and a review of their hypersensitivity. Contact Dennatitis33(2):100-5, 1995.
Smart ER, Macleod RI Lawrence CM Resolution of lichen planus following removal of amalgam restorations in patients with proven allergy to mercury salts: a pilot study. Br Dent J 178:108-2, 1995.
Suzuki N, Metal allergy in dentistry: detection of allergin metals with X-ray fluorescence spectroscope and its application toward allergin elimination. lnt J Prosthodont 8(4):351-69, 1995.
Copyright CDA Journal. Vol. 27, No. 4, April 1999.
Reprinted with permission